
Nobel Prize Series: Chemistry 2020
All About CRISPR
It used to be the subject of science fiction but now it’s as real as reality can get; you’ve read the title so you know what I’m talking about: the revolutionary technology of CRISPR. In fact, it was so revolutionary that it earned Emmanuelle Charpentier and Jennifer A. Doudna the Nobel Prize in Chemistry in 2020.
But what does CRISPR actually mean? What are its applications and drawbacks?
Let’s start at the beginning. At its core, CRISPR is a method for editing our genes — the small sections of DNA that code for certain features. For example, there are 16 genes associated with our eye colour; just like how certain coding languages help code a software, our genes make us us.
CRISPR helps us not only cut out certain genes but insert new ones too. This has endless applications in preventing or treating genetic diseases, creating more resistant crops, using bioenergy to increase our sustainability as a species, and much more. Before we explore more of these, let’s get into the nitty-gritty of CRISPR. Get ready to put your science hats on!
The Science Behind It All
Would you believe me if I told you that this amazing technology was first discovered as part of the defense mechanism of bacteria against viruses? Well it was! In as early as the 1980’s, scientists began noticing something interesting: there were short sequences of DNA that seemed to repeat every little bit in certain prokaryotes — that is bacteria and archaea — with different sequences interspaced in between.
Eventually, the term CRISPR was coined to refer to these short repeats, with CRISPR standing for
Clustered
Regularly
Interspaced
Short
Palindromic
Repeats
Step 1: Scientists eventually discerned that these CRISPR served a crucial purpose for bacteria: they would insert viral DNA that a virus inserted into them into the CRISPR section of their DNA.
Step 2: If the virus ever attacked this bacterium again, the CRISPR section of the bacterium’s DNA would be turned into guide RNA for a reason we’ll soon discuss.
Step 3: It would then bind to or attach to a protein designed to cleave or cut DNA — often this protein is CRISPR-associated protein 9 (or Cas9) hence why you may have heard of CRISPR being referred to as CRISPR Cas 9.
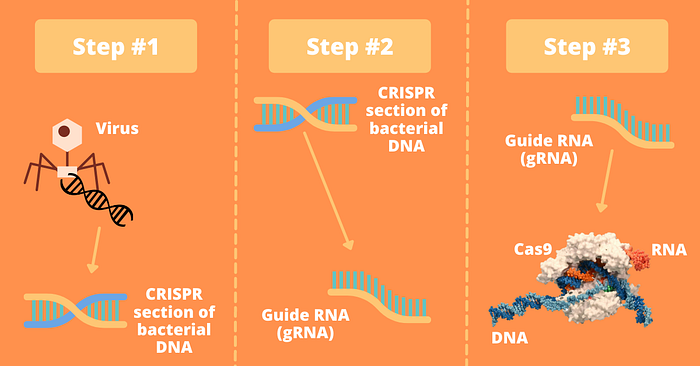
Let’s also quickly discuss some of the differences between DNA and RNA: DNA is how we store our genetic information and RNA is often how we move that information around. In order for information to move around, DNA is often transcribed into an RNA molecule which contains the exact same information as the DNA.
Step 4: Next, this complex of the guide RNA (gRNA) and Cas will search through the rest of the bacterium’s genes.
Step 5: If they find a matching DNA sequence to the gRNA, the Cas9 will cut that section of the DNA out, thereby destroying it. Boom, no more viral DNA. That is, except for the one that will be saved in the CRISPR section of the bacterium genome in case of a future attack.

The even cooler part? This “memory” the bacterium now has of the viral DNA is passed onto the bacterium’s progeny.
Let’s back up a second. If the viral DNA is also encoded in the bacterium’s DNA and the Cas9 complex is on the hunt for any DNA that matches that, how come the complex doesn’t end up cutting the bacterium’s own DNA? Phenomenal question. I have a two-word answer for you: PAM sequences.
PAM What?
The building blocks of our DNA are 4 molecules, or base pairs, called A, T, C, and G. PAM sequences are DNA sequences with 2–6 of these base pairs with two main roles in bacterial CRISPR systems…
- Ensure the Cas9 complex doesn’t cut and destroy the viral DNA intentionally placed in the CRISPR section of the bacterial DNA
- Speed up the “scanning” of DNA, since the complex only needs to check for the PAM sequence and if it matches, then it will go through the trouble of unwinding the DNA and seeing if it matches fully with the gRNA
How Do Things Differ When Using CRISPR for Gene Editing
The goal with CRISPR in bacteria is to disable the invading viral DNA and prevent the bacterium from dying. So cutting the viral DNA is enough. But in gene editing we can do something even more interesting.
Let’s break it up into two main categories: when the Cas9 cleaves the DNA, scientists can either let the DNA repair itself which often results in the cut gene being turned off, or introduce a new sequence of DNA and change the function of the gene entirely. Now that’s true editing.
Jennifer Doudna, co-inventor of CRISPR and Chemistry Nobel Prize laureate, likes to refer to the CRISPR system as molecular scissors that can make small, precise edits in the genome, much like how we can cut and paste words and letters in a word processing application.
Here’s another analogy that may help you better visualize the process: think of the whole Cas9 complex (including Cas9 and the gRNA) as a librarian who’s just found that a book has been misplaced.
The librarian’s eyes are like the gRNA, scanning the bookshelves and book spines, looking for the right place to put the misplaced book. Once their eyes — the gRNA — spots the incorrect book that’s taking the spot of the correct one, their hands — or the Cas9 — “cuts” the incorrect book out of that spot and inserts the correct one.
The only caveat with that analogy is that the DNA sequence that is cut with CRISPR isn’t necessarily “misplaced” or “incorrect” but sometimes it is.
Another way in which gene editing alters how CRISPR is used involves PAM sequences. When editing a genome, we don’t want the Cas9 complex to keep editing the same gene over and over as it recognizes the PAM. So, often scientists make a small change to the PAM sequence once the Cas9 complex has made the desired genetic edit. That way, the complex won’t mess up the experiment by editing the genome once more.
Are There Any Drawbacks?
Like all good things, CRISPR also has some problems, with one of the biggest ones being off-target effects.
This is when Cas9 complex goes elsewhere in the DNA, where it’s not supposed to and introduces breaks in the DNA. For obvious reasons, we don’t want that. Random changes in the DNA are never a good thing.
Another issue is on-target effects. These are when the complex edits the correct section of the DNA but it does so improperly. In other words, the repair at the location doesn’t work as it should.
Applications Galore
That brings us to the last topic we’ll discuss, some of the many applications of CRISPR in the real world.
The very first one, and possibly most important one, that comes to everyone’s mind is treating genetic diseases. One that’s been particularly focused on over the last few years has been sickle cell disease, the most common hereditary disease worldwide. This horrible disease affects hemoglobin, a protein crucial for normal red blood cells. By impairing hemoglobin, sickle cell disease results in a poor quality of life with patients experiencing frequent pain, damage to vital organs, lung problems, and more.

Since sickle cell affects so many people and is caused by one small mutation in the DNA, it makes a lot of sense for scientists to tackle it right now. And they’ve certainly done just that. Recently, the FDA gave the go ahead to run clinical trials in humans using CRISPR to treat sickle cell disease.
Another important, yet sometimes underappreciated, application is understanding the function of different proteins. Since CRISPR can disable certain genes, called knocking genes out, scientists can compare the differences between when the gene is turned on and off.
Some scientists are really thinking outside of the box and harnessing CRISPR in fascinating ways. For instance, they can attach enzymes to Cas9 that can increase or decrease the production of certain genes.
This is just a small taste of what CRISPR can do and our imagination is truly the only limit.
Interested in a video all about CRISPR? Check out this video from Nature that has beautiful 3D animations.
Time to Test Your Knowledge!
Now that you’ve learned lots about CRISPR, it’s time to see how much you remember. Complete the quiz below and see how you do.
About the Author
Parmin Sedigh is a 15-year-old stem cell and science communications enthusiast as well as a student researcher, based in Kingston, ON. She’s also the Director of Writing at Superposition and is working with the University of Guelph on a research project. You can usually find her on her computer following her curiosity. Connect with her on LinkedIn or email her at parminsedigh@gmail.com.
Sources
https://www.nature.com/articles/jhg2010126 https://www.nobelprize.org/uploads/2020/10/advanced-chemistryprize2020.pdf https://www.nobelprize.org/uploads/2020/10/chemistry-2020-figure2-en.pdf https://innovativegenomics.org/education/digital-resources/what-is-crispr/ https://youtu.be/4YKFw2KZA5o https://www.jax.org/personalized-medicine/precision-medicine-and-you/what-is-crispr# https://youtu.be/iSEEw4Vs_B4 https://blog.addgene.org/3-tips-for-improving-hdr-efficiency-for-editing-human-cells-with-crispr https://news.berkeley.edu/2021/03/30/fda-approves-first-test-of-crispr-to-correct-genetic-defect-causing-sickle-cell-disease/
